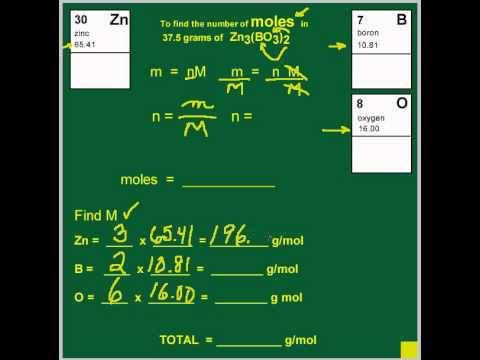
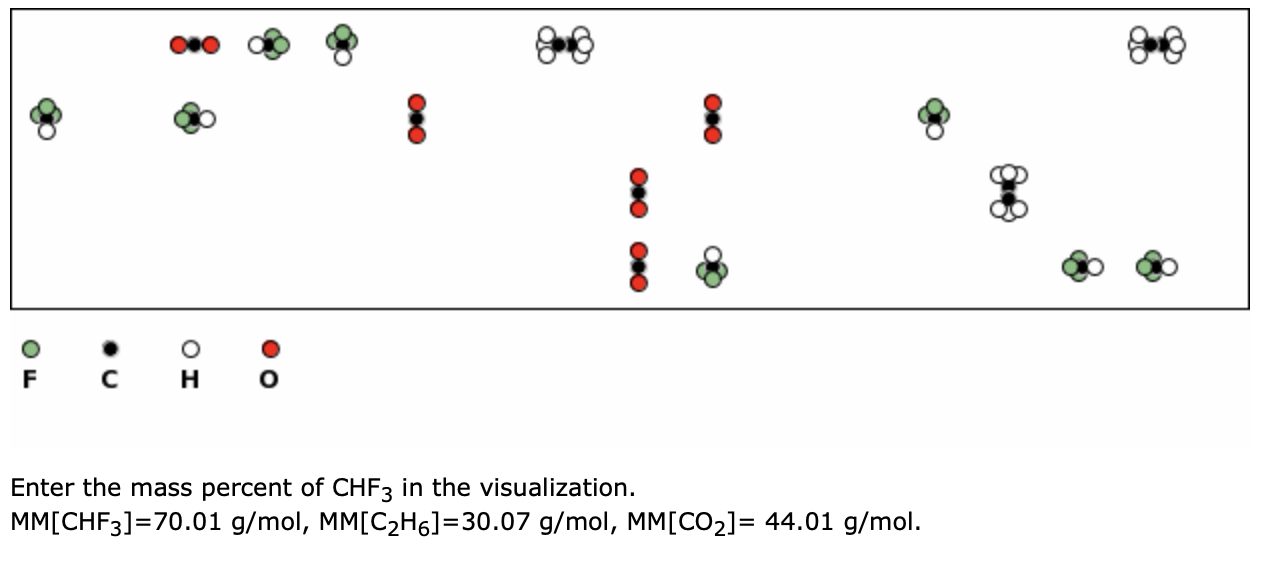
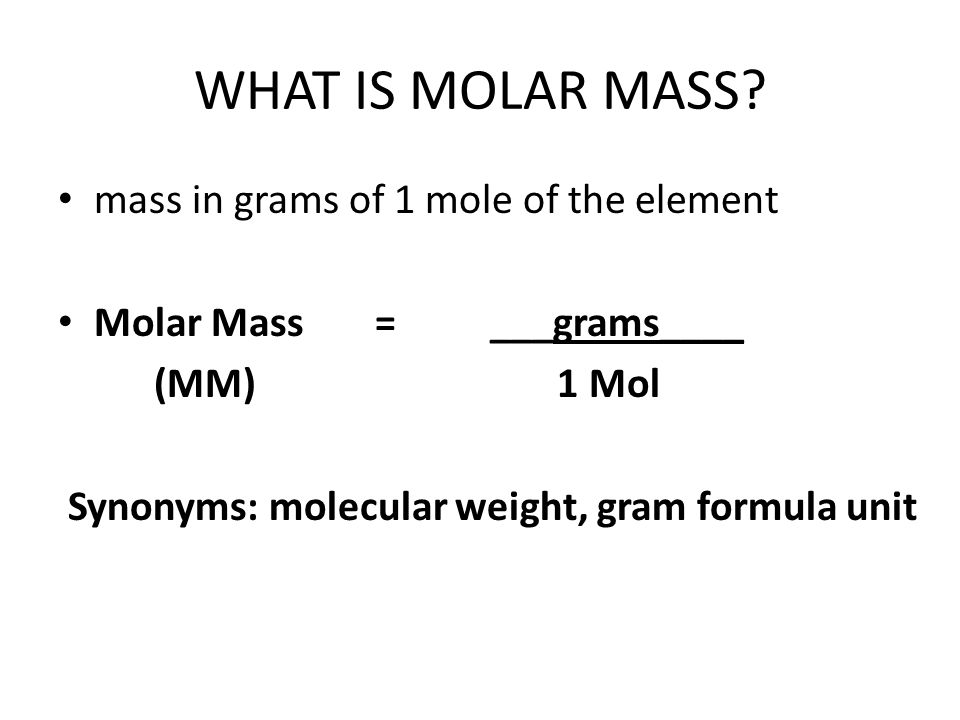Chapter 5 A Matter of Concentration. Ionic Phenomena = Things that happen to ions, which can be observed. - ppt download

SOLVED: If 50.0 g of CH₃OH (MM = 32.04 g/mol) are added to a 500.0 mL volumetric flask, and water is added to fill the flask, what is the concentration of CH₃OH

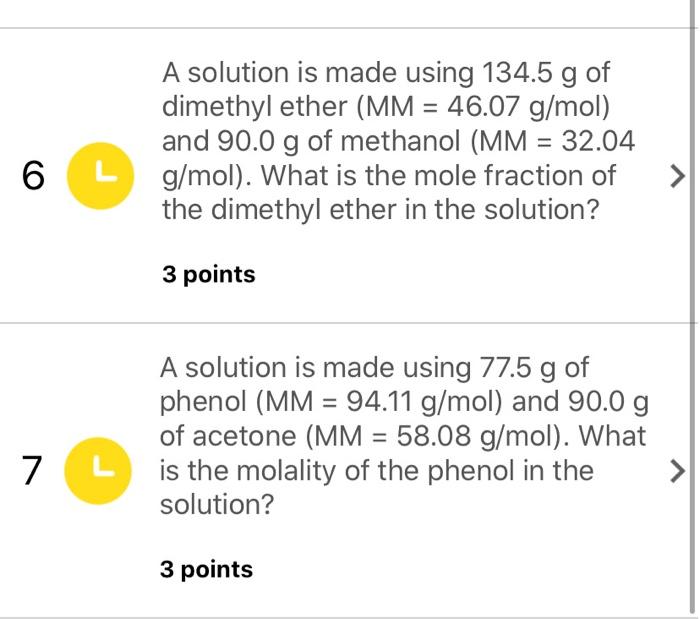
SOLVED: Question 21 ot 27 Submit What is the concentration in molarity of a solution which is 2.95 %mgv acetone (MM = 58.08 g/mol in ethanol (MM 46.07 g/mol)? x10

SOLVED: If 25.0 g of KBr (MM = 119.00 g/mol) are added to a 500.0 mL volumetric flask, and water is added to fill the flask, what is the concentration of KBr

Mole Practice 1. How many particles of gold are in 2.3 moles of Au? 2. Calculate the number of moles - Brainly.com

Molecular Dynamics and QM/MM Calculations Predict the Substrate-Induced Gating of Cytochrome P450 BM3 and the Regio- and Stereoselectivity of Fatty Acid Hydroxylation | Journal of the American Chemical Society

PLEASE HELP QUICKLY!!! MM H2O2 = 34.02 g/mol MM H2O = 18.02 g/mol MM O2 = 32 g/mol 2H2O2 —> 2H2O + - Brainly.com

mm-GBSA binding energy (ΔG, kcal/mol) of molecules to (A) DNMT1 and (B)... | Download Scientific Diagram

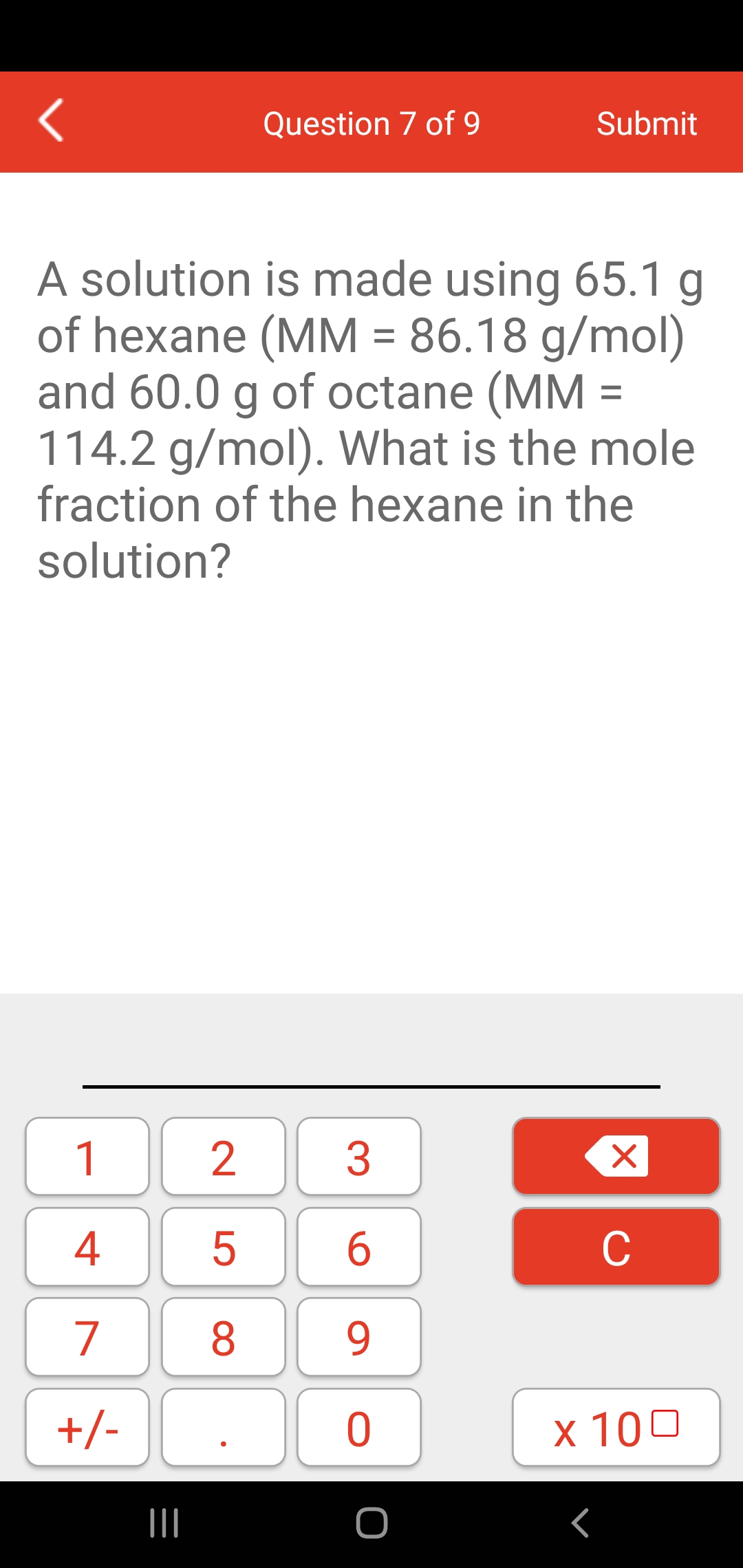
SOLVED: What is the concentration in molarity of a solution which is 11.39 %m/v octane (MM = 114.2 g/mol ) in hexane (MM = 86.18 g/mol)?