In three moles of ethane` (C_(2)H_(6))`, calculate the following: (i) Number of moles of carbon ... - YouTube
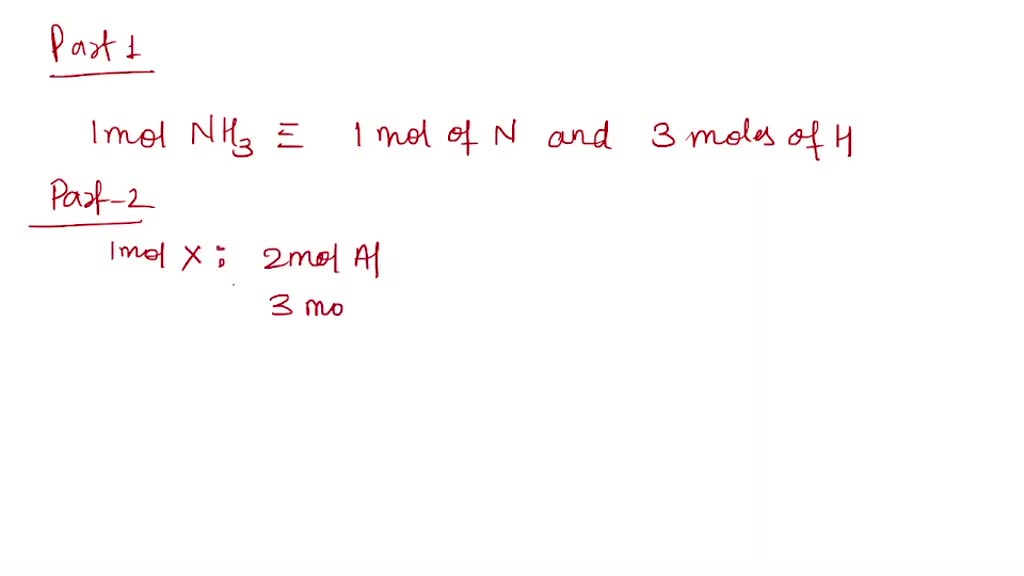
SOLVED: Part 1 (2 points) #d See Periodic Table See Hint 1mol NH3 contains mol Nand molH Part 2 (1 point) Write the chemical formula for 1 mol of compound X containing

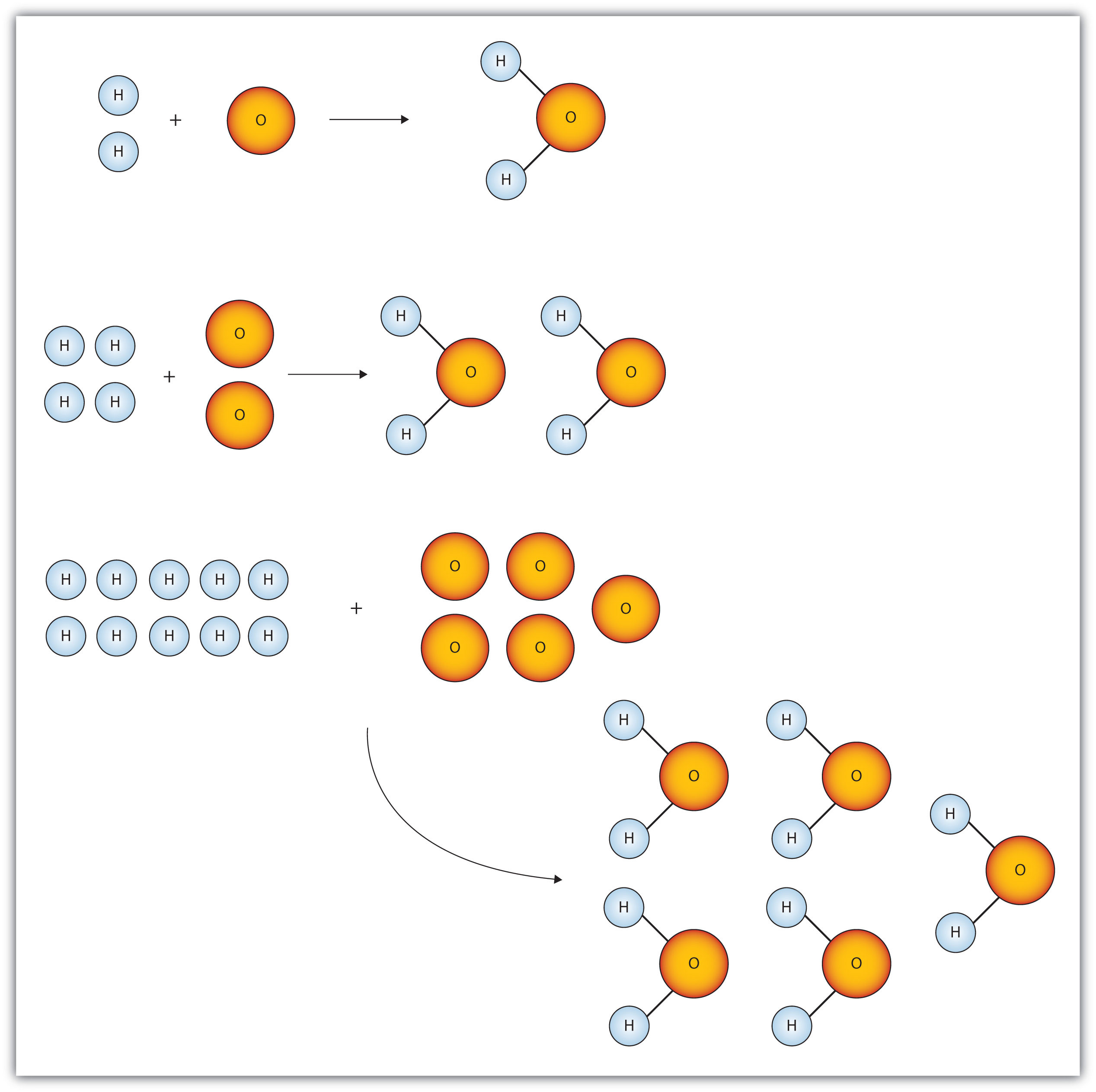
Chemistry Warm Up: Mole / Mass / Particles 1.What is the mass of one mole of water? 2.If one milliliter of water has a mass of 1.00grams, how many moles. - ppt download

STOICHIOMETRY CALCULATIONS COACH COX. MOLE TO MOLE CONVERSIONS Converting from moles of one substance in a chemical reaction to moles of another substance. - ppt download

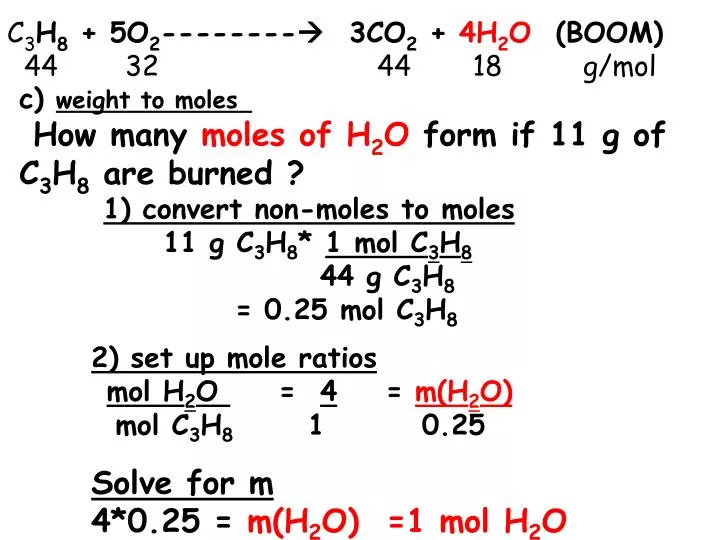
Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are
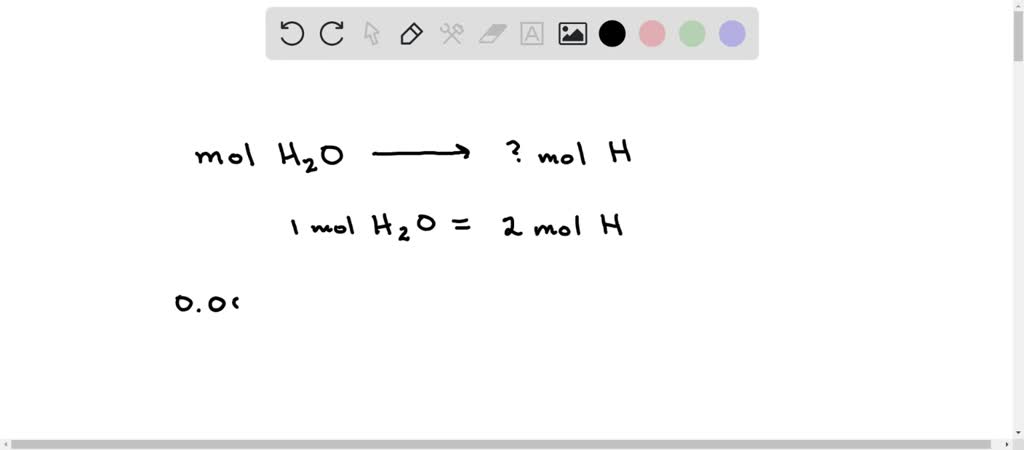
SOLVED: During the analysis, 0.00905 mol H2O is formed. Calculate the amount (mol) H in 0.00905 mol H2O.


![Solved Given that delta H^degree_f [H(g)] = 218.0 kJ.mol^-1 | Chegg.com Solved Given that delta H^degree_f [H(g)] = 218.0 kJ.mol^-1 | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F1a0%2F1a02aac8-2249-4506-869f-c847df3f3774%2FphpydBdke.png)