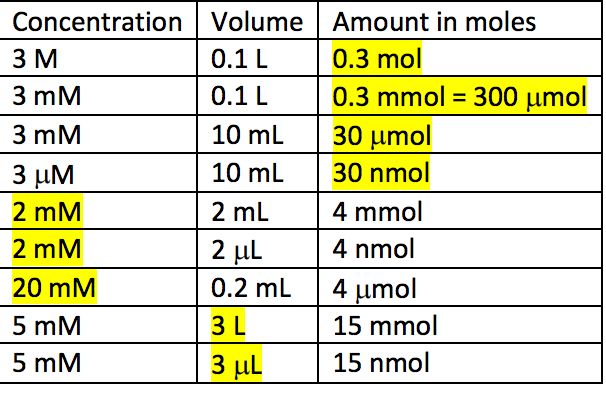
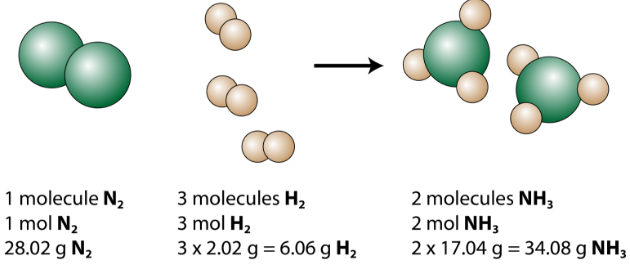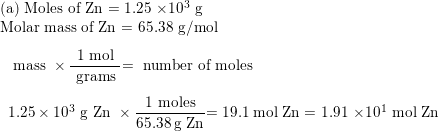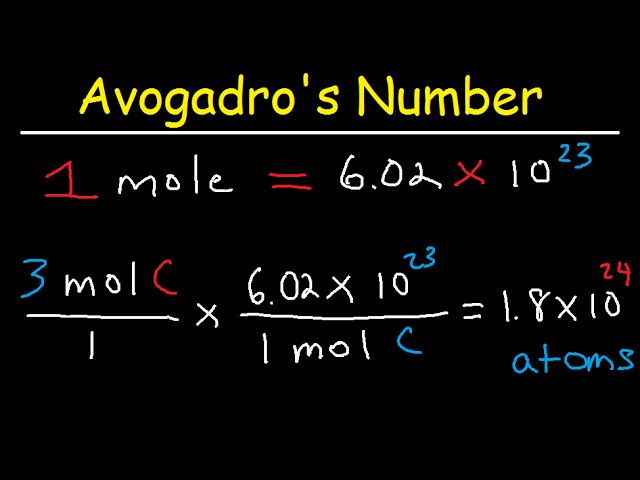Practice Problem How many moles of aluminum oxide will be produced from 0.50 mol of oxygen? 4 Al + 3 O 2 → 2 Al 2 O mol? mol 3 O 2 = 2 Al 2 O ppt download
Electrolyte KCl 3 mol/l | Maintenance and calibration solutions | Measuring Instruments | Labware | Carl Roth - International