
Dress, 19th century - Public domain dedication. Metropolitan Museum of Art image. - PICRYL - Public Domain Media Search Engine Public Domain Search

Alternative Splicing of a Receptor Intracellular Domain Yields Different Ectodomain Conformations, Enabling Isoform-Selective Functional Ligands - ScienceDirect

Pharmacological Inhibition of the Nod-Like Receptor Family Pyrin Domain Containing 3 Inflammasome with MCC950 | Pharmacological Reviews
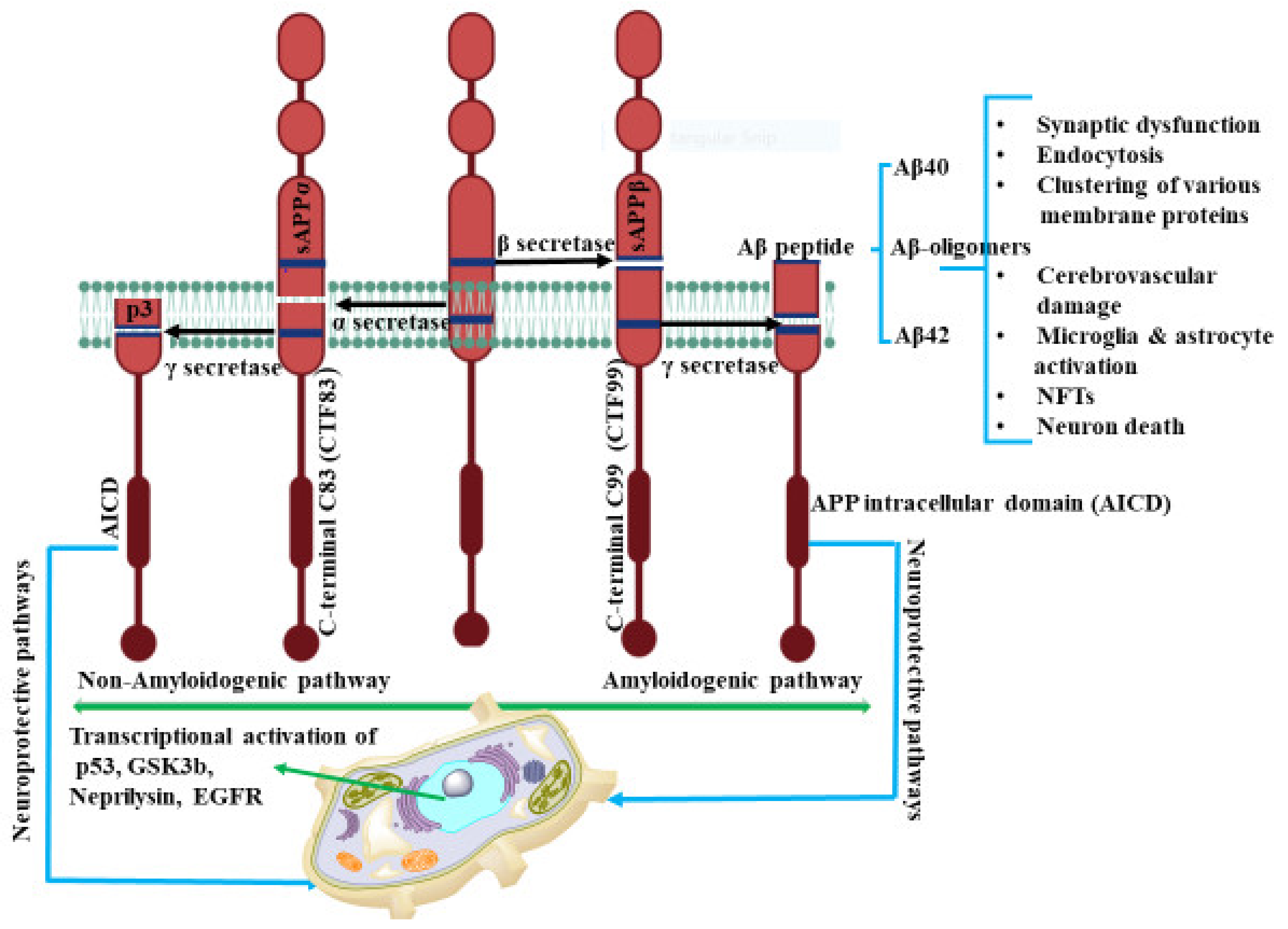
IJMS | Free Full-Text | Role of Receptors in Relation to Plaques and Tangles in Alzheimer’s Disease Pathology
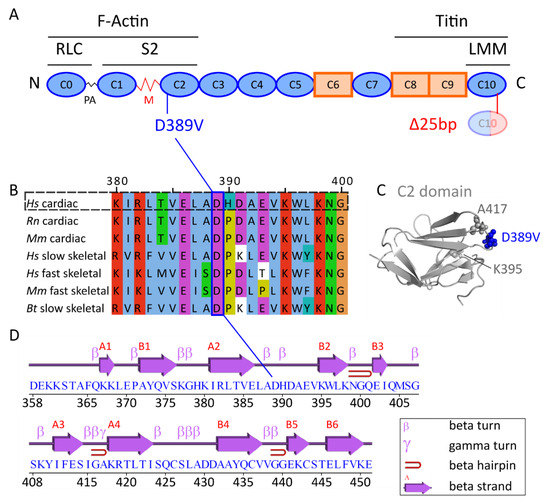
IJMS | Free Full-Text | Assessment of the Contribution of a Thermodynamic and Mechanical Destabilization of Myosin-Binding Protein C Domain C2 to the Pathomechanism of Hypertrophic Cardiomyopathy-Causing Double Mutation MYBPC3Δ25bp/D389V









