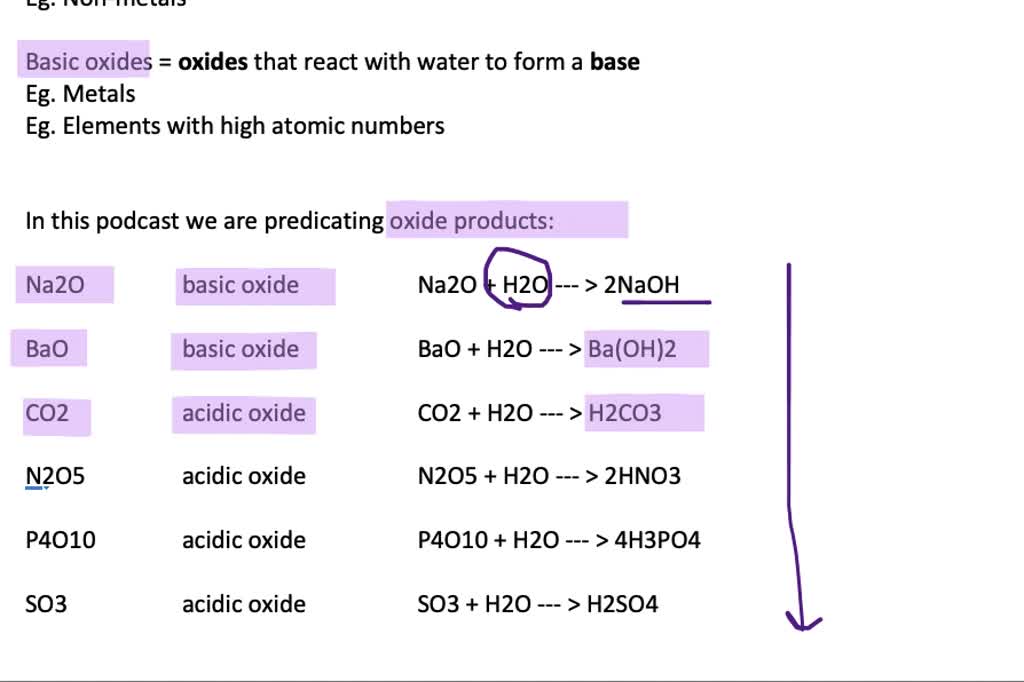
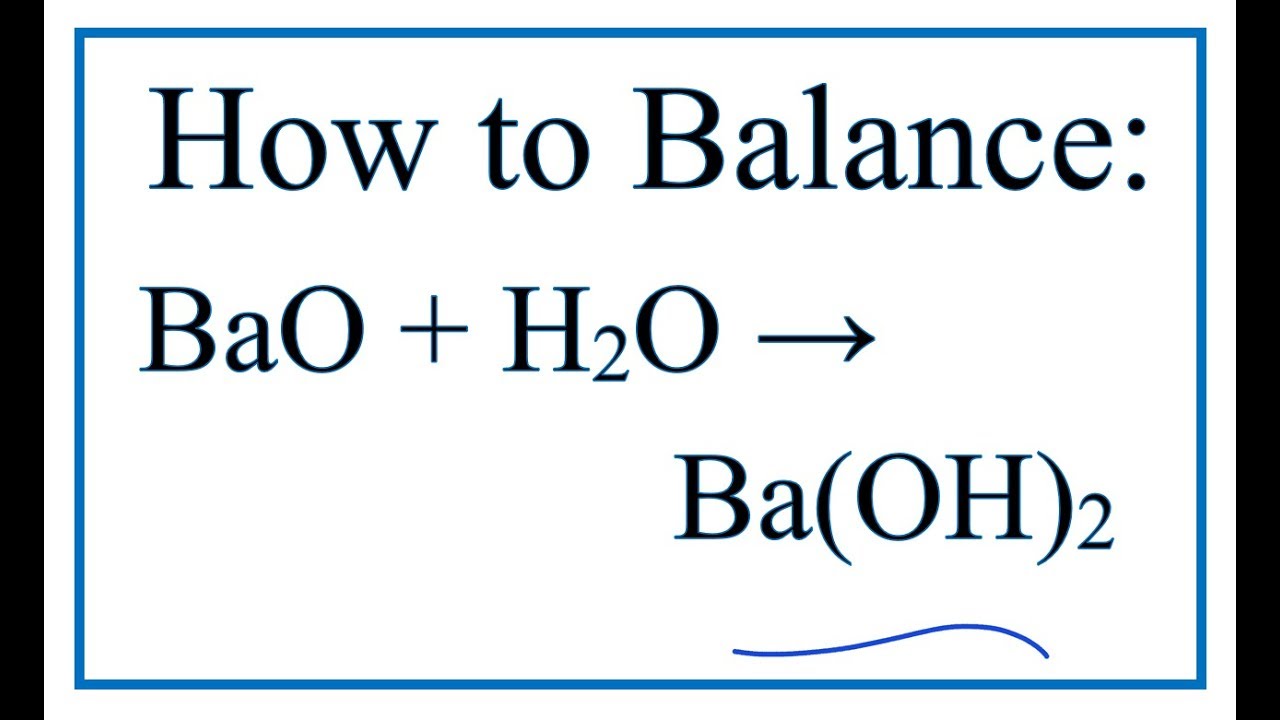
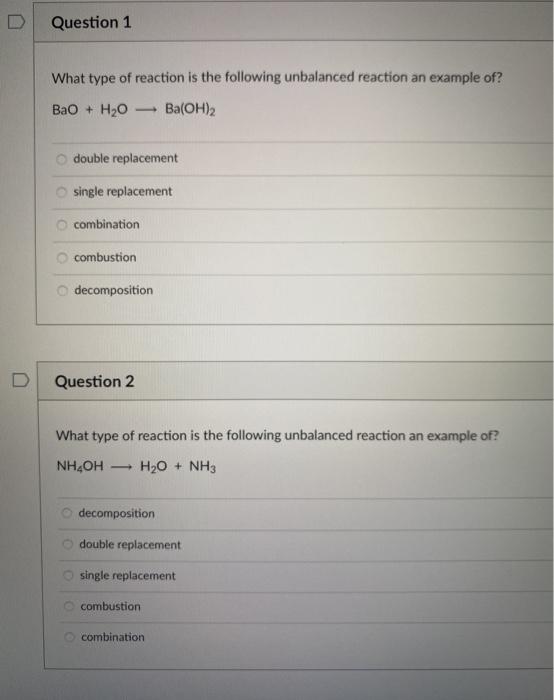
How to Balance BaO + H2O = Ba(OH)2 | BaO + H2O = Ba(OH)2 balance| Chemical equation BaO + H2O - YouTube
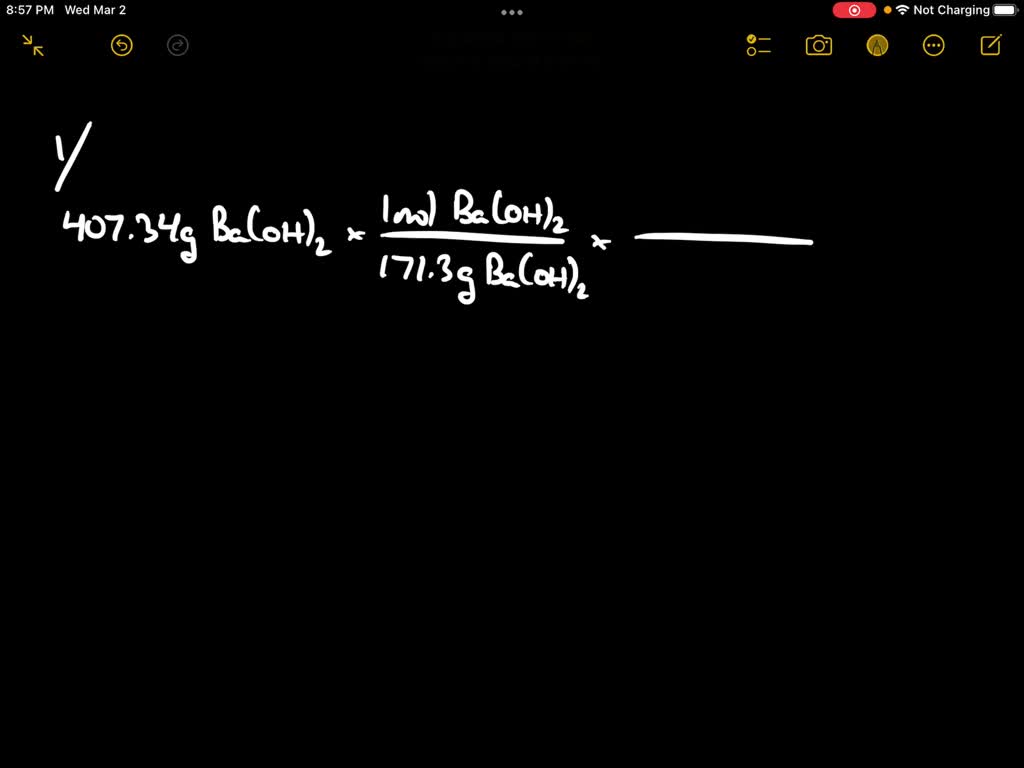
SOLVED: 1. BaO + H2O â†' Ba(OH)2 Given that you have: 407.34 grams Ba(OH)2 produced How many particles of BaO reacted? 2. For this reaction: K3PO4 + Al(NO3)3 â†' 3 KNO3 +