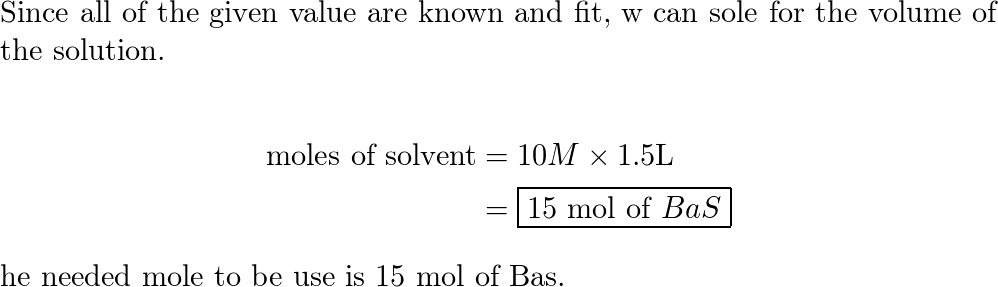For, A + B C , the equilibrium concentration of A and B at a temperature are 15 mol litre^-1 . When volume is doubted the reaction has equilibrium concentration of A

Nickel‐Catalyzed Thiolation of Aryl Nitriles - Delcaillau - 2021 - Chemistry – A European Journal - Wiley Online Library

SOLVED: if 0.40 mol of h2 and .15 mol of o2 were to reat as completely as possible to produce h20, what mass of the reactant would remain
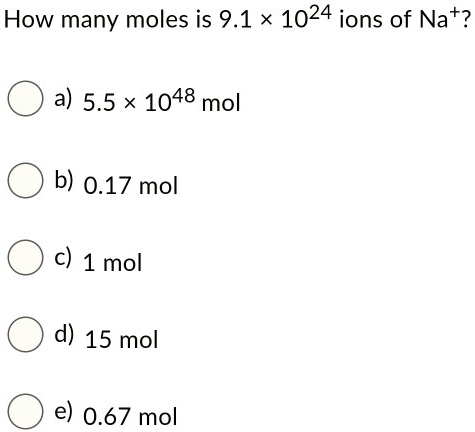
SOLVED: How many moles is 9.1 1024 ions of Nat? a) 5.5 * 1048 mol b) 0.17 mol 1 mol d) 15 mol e) 0.67 mol

SOLVED: Example: (1) How many lithium is 6.94 g/mole) moles are in 15 grams of lithium? (molar mass of 15 grams 1 mole 6.94 grams (2) How many 6.94grams mole) grams are

Cuántas moléculas hay en cada matraz? número de moléculas matraz vacío matraz con 15 mol de H2 - Brainly.lat
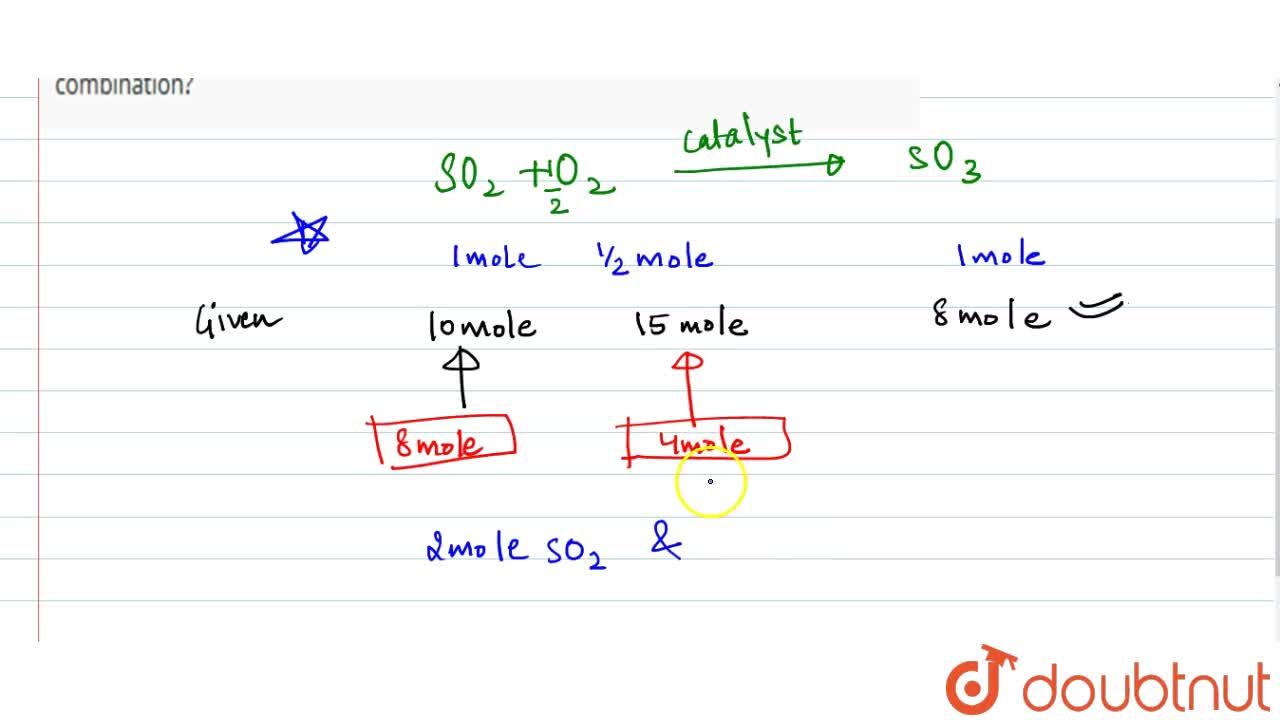
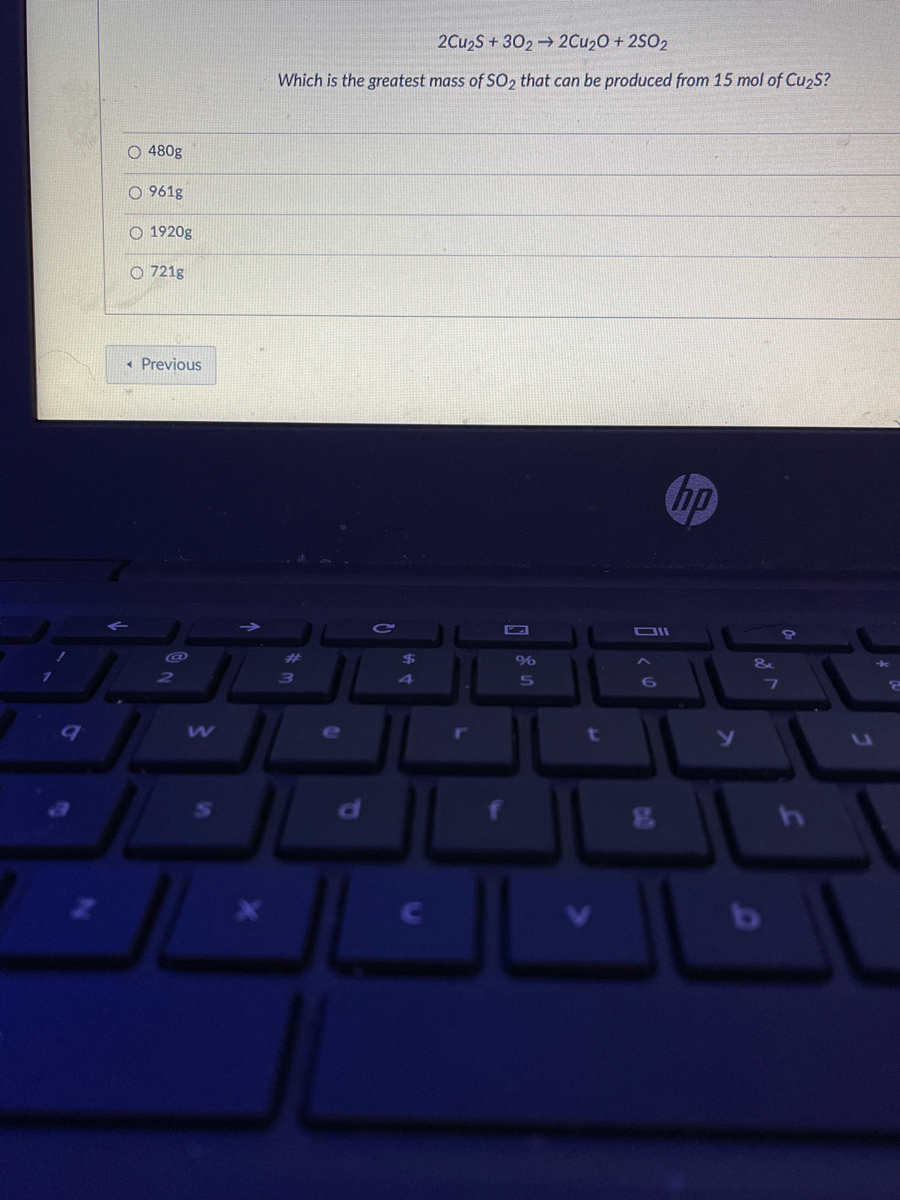
When a mixture of 10 moles of SO(2) and 15 moles of O(2) was passed over catalyst, 8 moles of SO(3) was formed. How many moles of SO(2) and O(2) did not
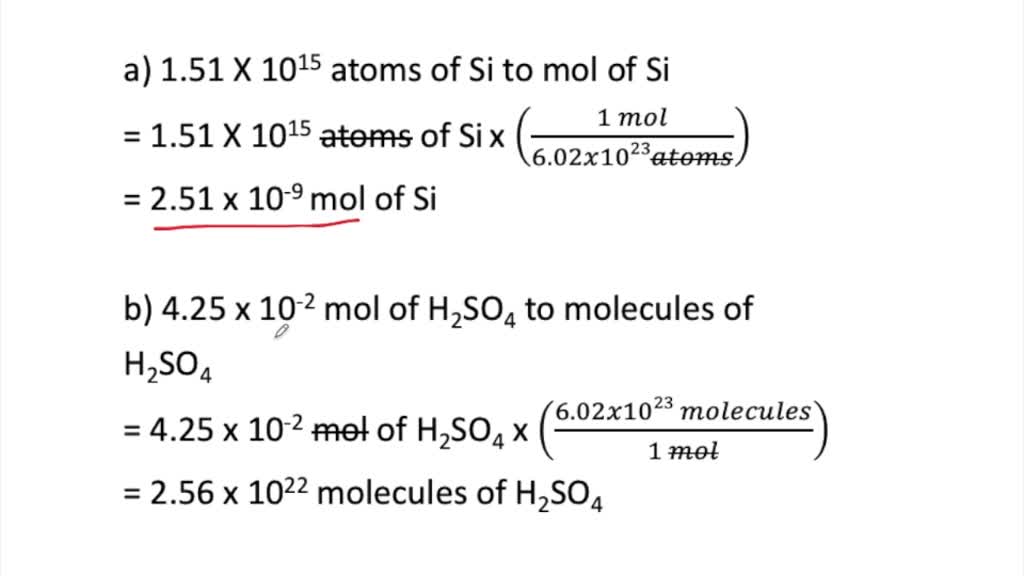
SOLVED:Perform the following conversions. a. 1.51 ×10^15 atoms of Si to mol of Si b. 4.25 ×10^-2 mol of H2 SO4 to molecules of H2 SO4 c. 8.95 ×10^25 molecules of CCl4

15 moles of N2 is mixed with 20 moles of H2 in an 8 litre vessel. 5.6 moles of ammonia is formed Calculate Kc for the equation, N2(g) +3 H2(g)= 2NH3(g) "+ heat"
15 moles of H2 and 5.2 moles of I2 are mixed and then allowed too attain equilibrium at 500^°C .At equilibrium the concentration of HI its found to be 10 moles .

りん酸緩衝剤粉末 (1/15 mol/L pH 7.4)・Phosphate Buffer Powder (1/15 mol/L, pH 7.4)・167-14491【詳細情報】|【ライフサイエンス】|試薬-富士フイルム和光純薬